Electron Orbitals
Schrödinger Equation
The Schrödinger equation is a partial differential equation that governs the wave function of a non-relativistic quantum-mechanical system. Its discovery was a significant landmark in the development of quantum mechanics. It is named after Erwin Schrödinger, an Austrian physicist, who postulated the equation in 1925 and published it in 1926, forming the basis for the work that resulted in his Nobel Prize in Physics in 1933.
Atomic Models
Bohr model
The Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic sh
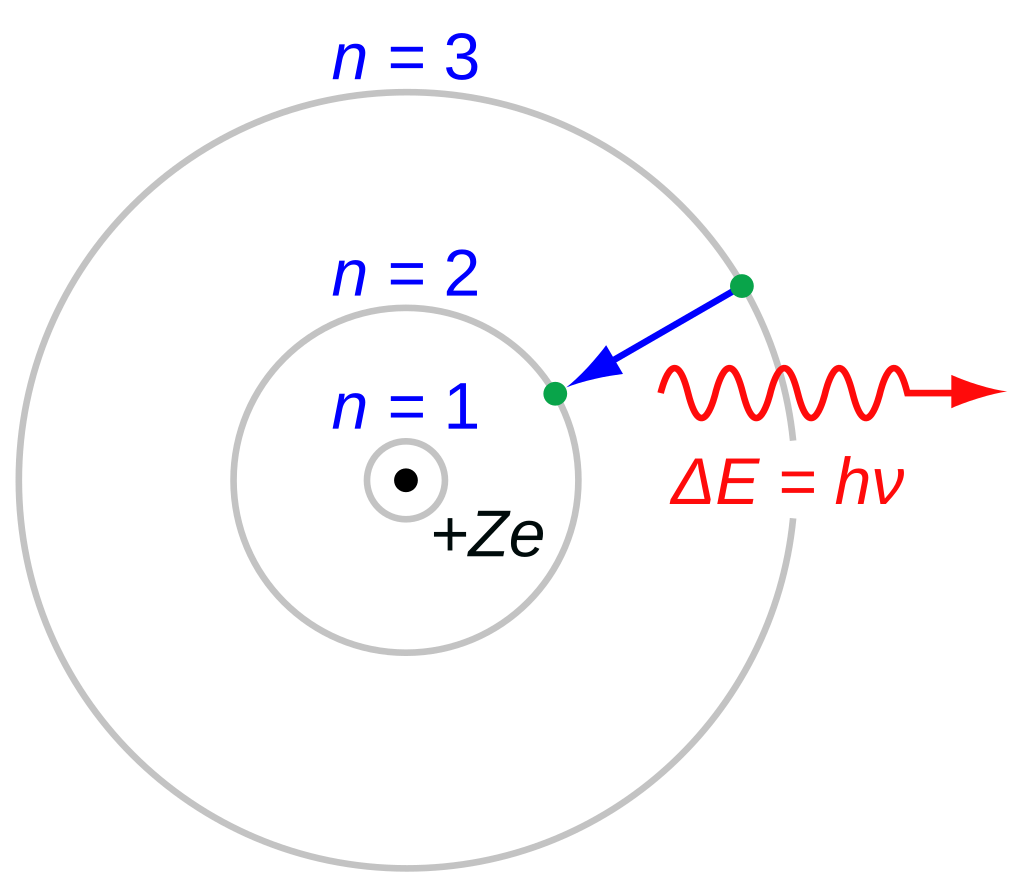
The Bohr model of the hydrogen atom (Z = 1) or a hydrogen-like ion (Z > 1), where the negatively charged electron confined to an atomic sh
encircles a small, positively charged atomic nucleus and where an electron jumps between orbits, is accompanied by an emitted or absorbed amount of electromagnetic energy (hν). The orbits in which the electron may travel are shown as grey circles; their radius increases as _n_2, where n is the principal quantum number. The 3 → 2 transition depicted here produces the first line of the Balmer series, and for hydrogen (Z = 1) it results in a photon of wavelength 656 nm (red light).
Kvantová Čísla
Kvantová čísla jsou čísla, kterými se v kvantové mechanice popisují vlastnosti určitých částic v systému; každé číslo odpovídá jedné zachovávané veličině. Nejčastějším použitím kvantových čísel je popis elektronů a jejich orbitalů v atomovém obalu, například v chemii.
Kvantové číslo je charakteristikou kvantového stavu.
Quantum Numbers
Schrödinger’s approach results in three quantum numbers that can be used to define an orbital (wave function), the principle (
Principal quantum number (
Hlavní kvantové číslo[2]
The principal quantum number (n) tells the average relative distance of an electron from the nucleus:
As n increases for a given atom, so does the average distance of an electron from the nucleus. A negatively charged electron that is, on average, closer to the positively charged nucleus is attracted to the nucleus more strongly than an electron that is farther out in space. This means that electrons with higher values of n are easier to remove from an atom. All wave functions that have the same value of n are said to constitute a principal shell because those electrons have similar average distances from the nucleus. As you will see, the principal quantum number n corresponds to the n used by Bohr to describe electron orbits and by Rydberg to describe atomic energy levels. As we will see, these can also be related to the periods of the periodic table.[1:1]
Azimuthal Quantum Number (
Vedlejší kvantové číslo[2:1]
The second quantum number is often called the azimuthal quantum number (
For example, if n = 1,
In naming orbitals we often denote the azimuthal number with a symbol.
Magnetic Quantum Number (
Magnetické Kvantové číslo[2:2]
The third quantum number is the magnetic quantum number (
For each value of
The allowed values of
For example, if
Each wavefunction with an allowed combination of
Spin Quantum Number (
Spinové Kvantové Číslo[2:3]
There is a fourth quantum number, which is the electron spin quantum number. The above three quantum numbers come from the solutions of the Schrödinger wave equation and describe the distance from the nucleus, the shape and orientation of the electronic orbitals that can exist for a hydrogen like atom. But there are actually two orbitals within each of these orbitals, and these result from the intrinsic spin of an electrons, which produces an intrinsic magnetic field. [3]
It should be noted that in 1928 Paul Dirac developed a relativistic wave equation, the Dirac Equation, that was consistent with both quantum mechanics and the theory of special relativity, and not only accounted for all four quantum numbers, but even introduced the concept of antimatter. [3:1]
Naming Orbitals
The first three quantum numbers define the geometric shapes of the orbitals and can be used to name them. The convention is to first indicate the principle quantum number (n), followed by a letter designating the azimuthal quantum number and then using a subscript to indicate the magnetic quantum number. The magnetic quantum number is often not specified. Note, the ml value may be the true values (-3,-2,-1,0,+1,+2,+3), or as we will see in the next section, expressions within the x,y,z Cartesian coordinate system.[3:2]

Number of each orbital
For each shell there are n orbitals. The following table shows the allowed orbitals for ground state configurations, for each shell (principle quantum number) you add an azimuthal orbital (the allowed values of l for each n are 0 to n-1), where l=0 is an s orbital, l=1 is a p, l=2 is a d....)[3:3]
As we shall see in the next Chapter, the structure of the periodic table is based on the filling of the orbitals in their lowest energy state, and the orbitals in orange (5g, 6g,6h,... are of such high energy that electrons do not occupy them if the electron is in a ground state configuration.[3:4]
Now because the are
n= 1 as one s orbital = (1 total)
n=2 has one s and three p (4 total)
n=3 has one s, three p and 5 d (9 total)
n=4 has one s, three p, 5 d and 7 f (16 total)
The Shape of Atomic Orbitals
In the previous section we learned that electrons have wave/particle duality, exist in orbitals that are defined by the Schrödinger wave equation that involves the complex coordinate system and imaginary numbers, and that they can be defined by quantum numbers n, l and ml. We use the Greek symbol psi (
So when we say
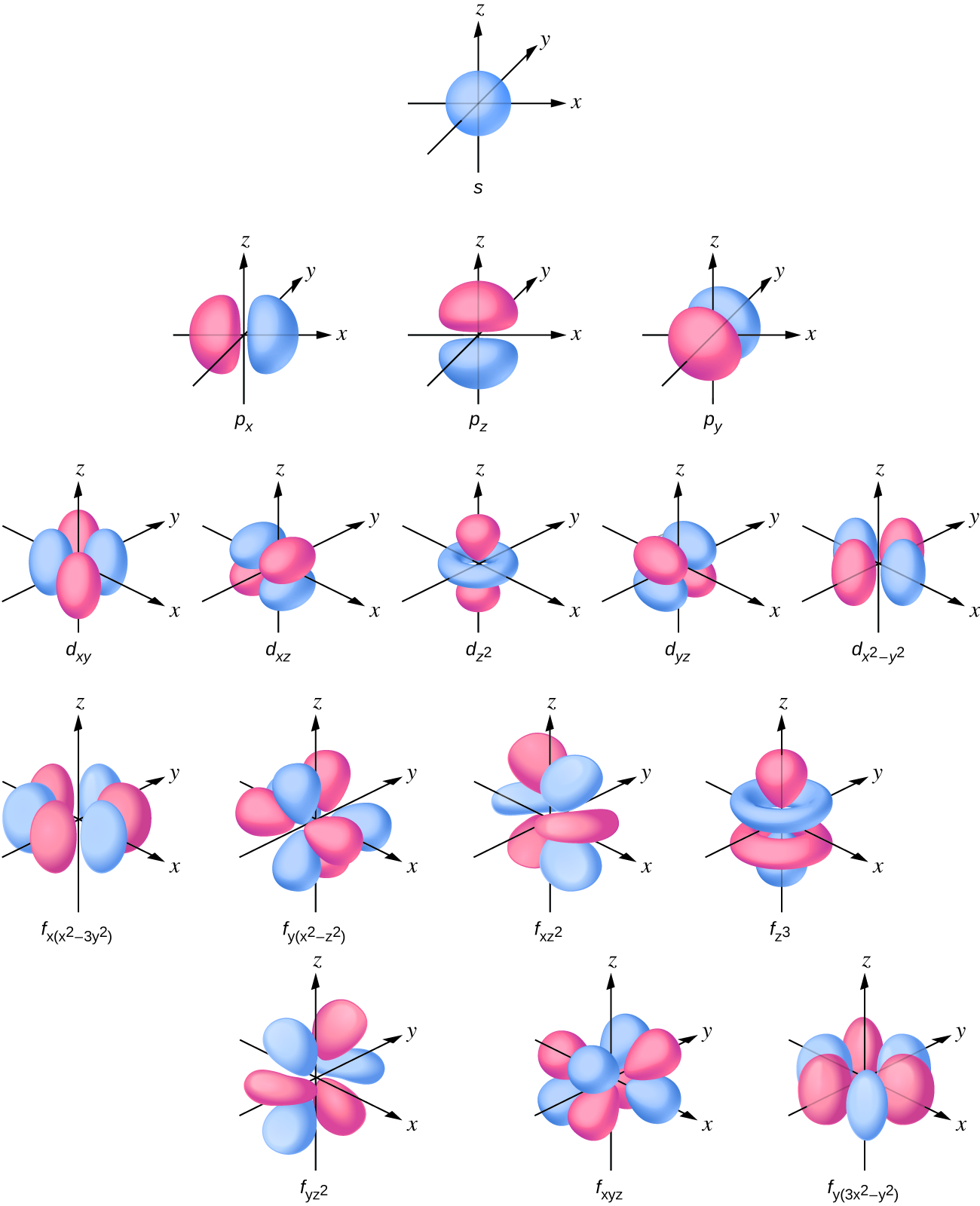
Figure 6.6.1: Select Cartesian coordinate visualizations of orbitals expressed in real space.
Note in Figure 6.6.1 that there is one type of s orbital
s-orbitals
For l=0 the electron density function is spherically symmetric and the 1s orbital has no nodes. Figure 6.6.2 shows the square of the wavefunction.
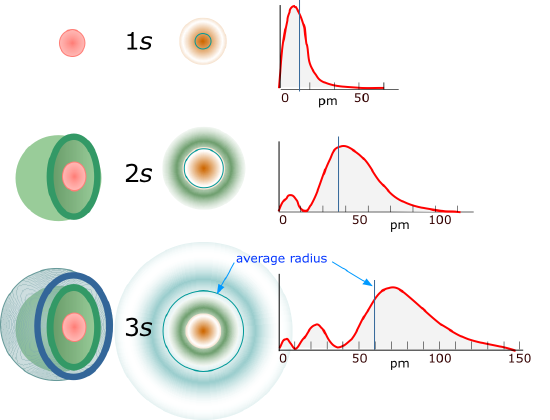
Figure 6.6.2: the radial distribution functions for the s orbitals of the first three principle quantum shells. This graph represents
As the principle quantum number (
p-orbitals
For the
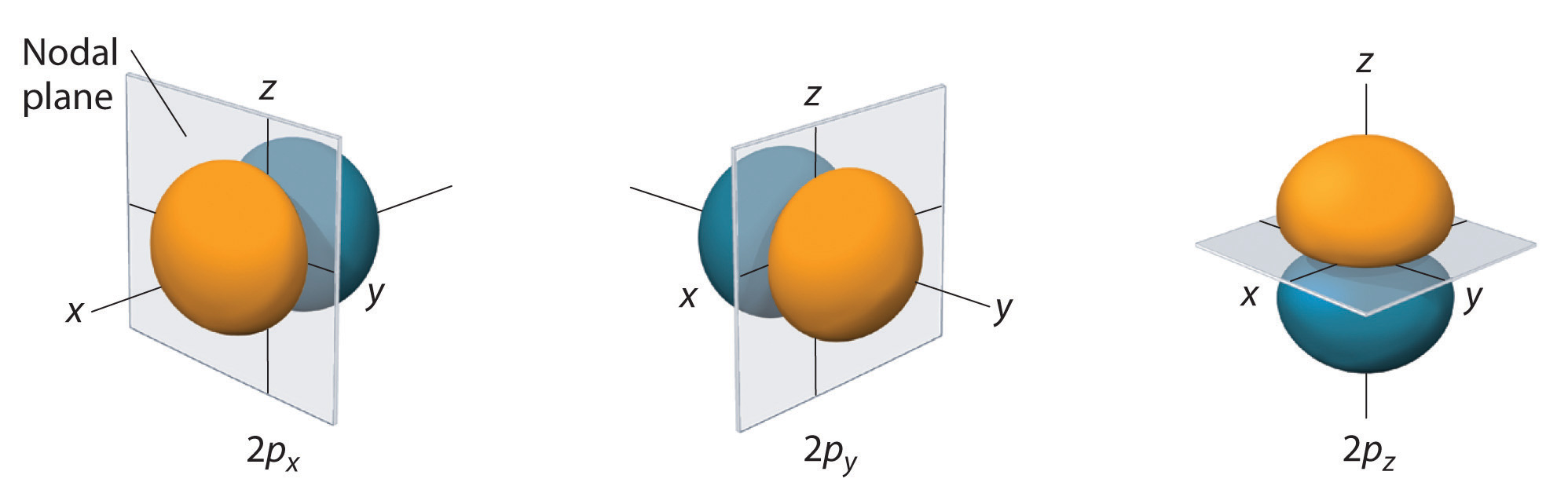
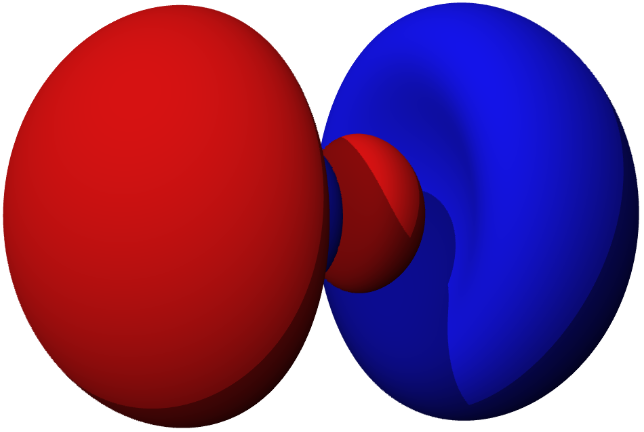
Figure 6.6.3: 2 p orbitals along the x, y and z axes (left) and a 3 p orbital (right).
Just as for s orbitals, as you move to higher principle quantum numbers the number of nodal surfaces increases, but they are no longer simple planar surfaces (see Figure 6.6.3). [4:2]
Practice
https://chem.libretexts.org/Courses/University_of_Arkansas_Little_Rock/Chem_1402%3A_General_Chemistry_1_(Belford)/Text/6%3A_The_Structure_of_Atoms/6.5%3A_The_Modern_View_of_Electronic_Structure#Quantum_Numbers ↩︎ ↩︎ ↩︎ ↩︎ ↩︎ ↩︎ ↩︎
https://cs.wikipedia.org/wiki/Kvantové_číslo#Elektron_v_atomu ↩︎ ↩︎ ↩︎ ↩︎
https://chem.libretexts.org/Courses/University_of_Arkansas_Little_Rock/Chem_1402%3A_General_Chemistry_1_(Belford)/Text/6%3A_The_Structure_of_Atoms/6.5%3A_The_Modern_View_of_Electronic_Structure#Naming_Orbitals ↩︎ ↩︎ ↩︎ ↩︎ ↩︎
https://chem.libretexts.org/Courses/University_of_Arkansas_Little_Rock/Chem_1402%3A_General_Chemistry_1_(Belford)/Text/6%3A_The_Structure_of_Atoms/6.6%3A_The_Shapes_of_Atomic_Orbitals ↩︎ ↩︎ ↩︎